Access Site Complications by Intracardiac Interventions
- Authors: Kimkov A.V.1
-
Affiliations:
- St.-Katharinen Hospital GmbH Frechen
- Issue: Vol 2, No 1 (2022)
- Pages: 5-12
- Section: Reviews
- URL: https://journals.eco-vector.com/cardar/article/view/100389
- DOI: https://doi.org/10.17816/cardar100389
- ID: 100389
Cite item
Abstract
In recent decades, the number of intracardiac procedures using percutaneous puncture access has increased manifold. Despite the acquisition by operators of expertise and standardization of methods, the problem of complications remains relevant.
AIM: to analyze the frequency and nature of complications of percutaneous access in intracardiac interventions. Suggest recommendations to reduce the incidence of complications.
MATERIALS AND METHODS: analysis of data published in international peer-reviewed journals on the topic, as well as the experience of the vascular surgery clinic of St. Katarina's Hospital.
CONCLUSIONS: the frequency and severity of complications depend on the experience of the operator, the size and frequency of changing the instrument, as well as compliance with the rules of preoperative diagnosis and postoperative management of the patient.
RECOMMENDATIONS: standardized preoperative preparation, careful planning of the intervention, analysis of the state of the access vessels, compliance with the rules of vessel puncture and competent performance of postoperative compression in combination with the use of suturing devices according to indications can reduce the frequency and severity of complications.
Keywords
Full Text
In 1929, the German urologist W. Forsmann inserted a urinary catheter into his cubital vein and pushed it in a central direction, and then took a radiographic image of his chest in a frontal view. This event was the first proven case of cardiac catheterization [1]. Subsequently, in 1956, he was awarded the Nobel Prize in Medicine. In 1953, the Swedish radiologist Seldinger proposed a vessel puncture technique for subsequent manipulations, named after him [2]. Finally, in the 1990s, Kiemenej and Laarman developed and implemented in clinical practice the technology of transradial percutaneous coronary intervention [3]. Currently, interventional technologies are the basis of many specialties.
In 2018, 220,000 coronary stenting interventions were performed in the Russian Federation, with approximately a 4-fold increase over 10 years. In the USA, 75,000 atrial ablations were performed in 2017 [4]. In all cases, manipulations were performed either through transradial or transfemoral access. Discussions about the advantages of one approach over the other ended after the publication of several studies, especially the RIVAL study [5]. At present, the radial approach is preferred in case of a suitable anatomy and in the absence of specific contraindications.
Despite the vast experience gained, complications of the access site during interventional procedures remain a major problem. In the 1990s, complications occurred in 6% of all patients after coronary interventions; 22%–25% of these cases required blood transfusion, and 21%–38% required surgery [6, 7]. Currently, with the accumulation of experience, the incidence of complications has significantly decreased (up to 1%–2%) [8–11], although it is still a concern given the huge number of interventions. Therefore, the mortality rate in patients who survived such a complication is 7.5% (1.1% without complications), the duration of hospital stay is twice longer, and the cost of inpatient treatment is almost two times higher ($ 9,583 vs. $ 18,350) [12].
The main complications of the access site are hematoma, ischemia of the access site in the limb dependent on the vessel, hemorrhage (including hemodynamically significant), false aneurysm requiring revision, vessel evulsion, and infection. Socioeconomic and psychological problems (staff distraction, psychological trauma to the patient, blocking the patient, and excessive use of resources) should also not be ignored. Factors that increase the risk of complications include female gender, age of 65 and older, body surface area greater than 1.62 m2, long duration of the procedure, high puncture site with transfemoral access, history of hemorrhagic diathesis, obesity, uncontrolled arterial hypertension, renal failure (creatinine level > 2 mg/d), use of glycoprotein IIB/IIIA inhibitors, and peripheral atherosclerosis [8–14].
Complications of the radial access
Occlusion of the radial artery is the most common complication with appropriate access. It occurs in 2%–18% of all transradial interventions14,15, and its occurrence depends largely on the surgeon’s experience, the method of compression and, most importantly, the quality of its application. According to studies, the course of this complication is usually asymptomatic, requiring no special treatment. The pathogenetic process is based on thrombogenesis in areas of the affected intima. Occlusion of the radial artery becomes symptomatic only in case of palmar anastomoses failure or in the presence of ulnar artery occlusion. In this situation, conducting an Allen test before the study will help prevent the development of a serious complication. This test is technically simple and does not require additional resources. In case of occlusion in both arteries, the patient is threatened with a higher risk of ischemia in the region dependent on these vessels and, in case of an unfavorable course, the development of necrosis and the need for amputation. In addition, perioperative injection of 5,000 IU heparin reduces the risk of vessel occlusion (71% vs. 4.3%). Factors that increase the risk of radial artery occlusion include excessive postoperative compression, repeated punctures, and large sluice diameters.
Non-occlusive lesions of the radial artery. This group includes, first, intimal hyperplasia and vascular remodeling. Studies of postoperative intima using intravascular ultrasound scan showed significant intimal hyperplasia with a resulting significant reduction in the diameter of the latter. A small series using optical coherence tomography revealed intimal tears in a significant group of patients (67%) and media dissection (36%). This damage leads to a decrease in the quality of the vessel during its subsequent use as a graft in arterial myocardial revascularization. A retrospective series revealed a decrease in the early patency of such grafts.
Spasm of the radial artery occurs in 5%–10% of all interventions. It rarely leads to serious consequences; however, it can lead to the failure of the procedure. Risk factors include small vessel diameter, female gender, multiple catheter replacements, use of a long sluice, and lack of experience of the operating surgeon. The radial artery has a well-defined tunica media, which is under the control of α-1 adrenoreceptors. For prophylactic purposes, sedation of the patient and local anesthesia can be used to control the effect of circulating endogenous catecholamines. Spasms respond well to treatment. Currently, nitroglycerin, subcutaneous injections of lidocaine, and various vasodilatory cocktails are used. The use of low-profile hydrophilic sluices also contributes to a reduction in the risk of spasm [25, 26]. Ouadhour et al. suggested based on their own research that subcutaneous administration of 0.5 mg isosorbide dinitrate with 1% lidocaine for local anesthesia can improve the radial access function [27].
Vessel perforation is a rare complication, which can lead to serious consequences in the form of loss of access, the need for conversion and, in case of an untimely response, the development of hematoma, compartment syndrome, and ischemia of the dependent area. In an analysis by Calvino-Santos et al., its incidence of less than 1% of cases was reported. Data from a large series showed an incidence of < 0.1% [6]. Moreover, most of these complications occurred in short women with tortuous vessels of the forearm [30]. Access safety in these patients can be improved by using a long sluice [30] and a guiding catheter [31]. If perforation is detected early, a small-diameter peripheral balloon can be used for angioplasty [32].
Compartment syndrome is a much rare but hazardous complication that requires emergency fasciotomy. Its incidence has been described in large series of cases at 0.004% [33]. The diagnosis is established based on clinical manifestations in the form of a massive tumor of the forearm with symptoms of peripheral ischemia, primarily neurological disorders.
False aneurysm is another rare complication. It occurs in < 0.1% of cases [6] and manifests itself as a penetrating injury to the vessel, which was not recognized in due time. The false aneurysm course is also facilitated by multiple punctures of the vessel and incorrect application of compression of the access site (for example, TR-Band) after the end of the procedure. Diagnosis is made by ultrasound. Conservative (adequate compression, injection of thrombin into the false aneurysm cavity under ultrasound control [34, 35]) or surgical (revision of the access site with hematoma evacuation and suturing) treatment is performed. Ligature of the radial artery is rarely required.
Table 1 presents the most common complications of radial access, their incidence, and methods of prevention and treatment.
Table 1. Complications of transradial access (modified from Kanei)
Complication | Incidence | Risk factors | Prevention and treatment |
Occlusion | 2%–18% | Overcompression Repeated punctures Large sluice diameter | Anticoagulation Quality hemostasis |
Non-occlusive involvement of the radial artery | often | Critical evaluation when used as a graft | |
Hand ischemia | Very rare | Prolonged cannulation | Pre-procedure assessment of the circulation |
Spasm | 5%–10% | Small vessel diameter Female gender Multiple catheter replacements Large sluice diameter Insufficient experience | Antispasmodic therapy Sparing manipulation |
Perforation | 0.1% | Aggressive manipulation Excessive anticoagulation | Timely diagnosis and bandaging |
False aneurysm | <0.1% | Multiple punctures Bacterial contamination of the catheter Excessive anticoagulation Large sluice diameter | Compression Thrombin injection Bandaging |
Nerve involvement | Extremely rare | Multiple punctures | Neurological therapy |
A-B fistula | Extremely rare | Numerous punctures | Surgical correction if necessary |
Significant bleeding | 0.15% | Correct hemostasis Transfusion Surgical treatment if necessary |
Complications of the transfemoral access
Despite the proven advantages of the transradial approach, the transfemoral approach continues to be widely used when indicated or preferred by a particular operating surgeon. The most common complications of this approach are puncture site hematoma, retroperitoneal hematoma, false aneurysm, arteriovenous fistula, and arterial dissection restricting the blood flow. Less complications are infections, thrombosis, and long-term lesions of nerve structures. Incorrect puncture of the vessel plays a huge role in the occurrence of these complications. Many interventional specialists do not have practical surgical experience and often deviate from the recommended puncture technique, considering it an insignificant stage of the intervention.
In addition, the anatomy of the common femoral artery is characterized by low variability expressed in different bifurcation heights. Ultrasound-guided puncture is generally recommended. When performing a puncture under radiological control, a specialist should focus on the femoral head and puncture slightly medially to its center toward the xiphoid process at an angle of 45°. The anatomy of the common femoral artery is schematically presented in Figure 1.
Fig. 1. Topography of the femoral artery (from [36])
Puncture should not be performed above the inguinal ligament, since in this area, the external iliac artery is oriented in the anteroposterior direction, and the puncture cannula passes parallel to the vessel without providing a puncture. In addition, in case of a high puncture, there is no bone base for performing postoperative compression. In this regard, the risk of bleeding with the formation of a retroperitoneal hematoma increases significantly.
The following factors should be considered risk factors for complications:
- insufficient knowledge of the femoral artery anatomy;
- little experience of the operating surgeon;
- increased BMI;
- female gender;
- refusal of ultrasonic navigation;
- agitated patient.
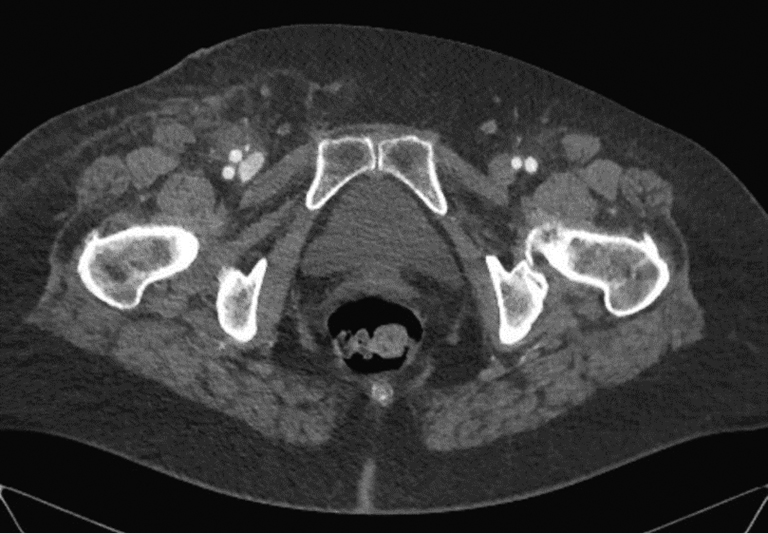
Hematoma of the puncture site is the most common complication, and it has been reported to occur in 5%–23% of cases by different authors. The hematoma scope varies from slight staining of the skin at the puncture site to a massive tumor requiring surgical treatment and blood transfusion. First, the causes of hematoma include multiple puncture attempts, incorrect or insufficiently long-lasting postoperative compression, and incorrect use of suturing devices. In the vast majority of cases, hematomas do not require special treatment and persist for 1–2 weeks. Follow-up and local therapy with heparin ointment are usually sufficient. If a growing hematoma is detected during the intervention itself, it is possible to use endovascular methods of treatment, such as balloon compression or implantation of a short stent or stent prosthesis. In this case, a contralateral approach is required. If it becomes necessary to implant a stent or a stent graft, it should be taken into account that the puncture site is anterior to the hip joint, that is, it is located in the mobile segment. The implantation of a stent in such a segment poses the risk of implant fracture and, as a consequence, the occurrence of vascular obstruction, limiting blood flow. Sometimes surgical treatment is necessary due to the hematoma size. The CT presentation of such a hematoma is shown in Figure 2. In case of the rapid development of a massive hematoma, necrotic changes in soft tissues are also possible. An example of such a case is presented in Figure 3. A special variant of local hematoma leads to the occurrence of a false aneurysm (0.5%–9%), and emergency surgery is indicated in this case.When performing postoperative compression, the size of the tool used to catheterize the vessel should be considered. The compression force should be approximately 20 mmHg above the systemic systolic pressure. The reference points in Table 2 can be used to determine the compression time. When using tools larger than 6 Fr in diameter for arterial access, the use of a suturing device is recommended. In the early postoperative period, strict adherence to bed rest by the patient and control of the puncture site by the department staff are of key importance.
Fig. 2. Massive hematoma in the right inguinal region and thigh after percutaneous coronary intervention (author’s case)
Fig. 3. Massive puncture site hematoma extending to the proximal femur; a ― radiological image; b ― rapid soft tissue necrosis due to hematoma pressure from the inside; c ― clinical presentation 8 days after hematoma evacuation and revision of the puncture site (author’s case)
Table 2. Recommended compression and bed rest times for transfemoral arterial access
Tool | Compression time, min | Bed rest, h |
Catheter 4F | 5 | 2–4 |
Sluice 4F Catheter 5F | 10–15 | 6–8 |
Sluice 5F | 15–20 | 6–8 |
Sluice 6F | >20 | >8 |
Retroperitoneal hematoma is a rare but serious complication. It has an incidence of 0.8%–0.44%. The occurrence of this pathology is associated primarily with suprainguinal puncture of the external iliac artery. The main danger consists in the delayed manifestation of symptoms, namely hypovolemia, pain in the ipsilateral hypogastrium, and deterioration in the general condition of the patient. Diagnosis is based on physical examination, ultrasound scan, and CT scan with contrast agent. Treatment includes an emergency revision with the imposition of a vascular suture, hematoma evacuation, and drainage in the retroperitoneal space. In some cases, interpolate implantation and blood transfusion are required. Rebleeding or lymphocele occurs in rare cases (Figure 4).
Fig. 4. Giant lymphocele (> 2.5 L) after evacuation of a retroperitoneal hematoma (author’s case)
Arteriovenous fistula (0.2%–2.1%) occurs when an artery is accidentally punctured through a vein or when a vein is accidentally punctured through an artery (Figure 5). In most cases, such fistulas close spontaneously within a few weeks. Surgical treatment is indicated in case of signs of overload of the heart compartments.
Fig. 5. Post-intervention arteriovenous fistula. The arrow indicates contrasting of the femoral vein (author’s case)
To take home
- Before puncturing the radial artery, the Allen test should be performed;
- Control the tool position in the vessel lumen;
- Avoid frequent, unnecessary tool changes;
- Perform adequate hemostasis;
- When using suturing devices, follow the instructions for use;
- Perform manual compression properly;
- Encourage patients to stay in bed;
- Control the access site before discharge;
- Share the experience.
ADDITIONAL INFORMATION
Competing interests. The author declare that they have no competing interests.
Funding source. This study was not supported by any external sources of funding.
About the authors
Alexander V. Kimkov
St.-Katharinen Hospital GmbH Frechen
Author for correspondence.
Email: alexandre04@inbox.ru
ORCID iD: 0000-0002-1774-938X
doctor of medical sciences
Germany, GermanyReferences
- Radner S. Thoracal aortography by catheterization from the radial artery; preliminary report of a new technique. Acta radiol. 1948;29(2):178–180. doi: 10.3109/00016924809132437
- Seldinger SI. Catheter replacement of the needle in percutaneous arteriography; a new technique. Acta radiol. 1953;39(5):368–376. doi: 10.3109/00016925309136722
- Kiemeneij F, Laarman GJ. Percutaneous transradial artery approach for coronary stent implantation. Cathet Cardiovasc Diagn. 1993;30(2):173–178. doi: 10.1002/ccd.1810300220
- Campeau L. Percutaneous radial artery approach for coronary angiography. Cathet Cardiovasc Diagn. 1989;16(1):3–7. doi: 10.1002/ccd.1810160103
- Mansour M, Karst E, Heist EK, et al. The Impact of First Procedure Success Rate on the Economics of Atrial Fibrillation Ablation. JACC: Clinical Electrophysiology. 2017;3(2):129–138. doi: 10.1016/j.jacep.2016.06.002
- Byrne RA, Cassese S, Linhardt M, Kastrati A. Vascular access and closure in coronary angiography and percutaneous intervention. Nat Rev Cardiol. 2013;10:27–40. doi: 10.1038/nrcardio.2012.160
- Waksman R, King SB III, Douglas JS, et al. Predictors of groin complications after balloon and new-device coronary intervention. Am J Cardiol. 1995;75(14):886–889. doi: 10.1016/s0002-9149(99)80681-x
- Omoigui NA, Califf RM, Pieper K, et al. Peripheral vascular complications in the Coronary Angioplasty Versus Excisional Atherectomy Trial (CAVEAT-I). J Am Coll Cardiol. 1995;26(4):922–930. doi: 10.1016/0735-1097(95)00263-4
- Piper WD, Malenka DJ, Ryan TJ Jr., et al. Predicting vascular complications in percutaneous coronary interventions. Am Heart J. 2003;145(6):1022–1029. doi: 10.1016/S0002-8703(03)00079-6
- Dauerman HL, Applegate RJ, Cohen DJ. Vascular closure devices: the second decade. J Am Coll Cardiol. 2007;50(17):1617–1626. doi: 10.1016/j.jacc.2007.07.028
- Marso SP, Amin AP, House JA, et al. Association between use of bleeding avoidance strategies and risk of periprocedural bleeding among patients undergoing percutaneous coronary intervention. JAMA. 2010;303(21):2156–2164. doi: 10.1001/jama.2010.708
- Tavris DR, Gallauresi BA, Lin B, et al. Risk of local adverse events following cardiac catheterization by hemostasis device use and gender. J Invasive Cardiol. 2004;16(9):459–464.
- Romaguera R, Wakabayashi K, Laynez-Carnicero A, et al. Association between bleeding severity and long-term mortality in patients experiencing vascular complications after percutaneous coronary intervention. Am J Cardiol. 2012;109(1):75–81. doi: 10.1016/j.amjcard.2011.08.007
- Sanmartin M, Gomez M, Rumoroso JR, et al. Interruption of blood flow during compression and radial artery occlusion after transradial catheterization. Catheter Cardiovasc Interv. 2007;70(2):185–189. doi: 10.1002/ccd.21058
- Pancholy SB. Transradial access in an occluded radial artery: new technique. J Invasive Cardiol. 2007;19(12):541–544.
- Spaulding C, Lefevre T, Funck F, et al. Left radial approach for coronary angiography: results of a prospective study. Cathet Cardiovasc Diagn. 1996;39(4):365–370. doi: 10.1002/(SICI)1097-0304(199612)39:4<365::AID-CCD8>3.0.CO;2-B
- Sakai H, Ikeda S, Harada T, et al. Limitations of successive transradial approach in the same arm: the Japanese experience. Catheter Cardiovasc Interv. 2001;54(2):204–208. doi: 10.1002/ccd.1268
- Saito S, Ikei H, Hosokawa G, Tanaka S. Influence of the ratio between radial artery inner diameter and sheath outer diameter on radial artery flow after transradial coronary intervention. Catheter Cardiovasc Interv. 1999;46(2):173–178. doi: 10.1002/(SICI)1522-726X(199902)46:2<173::AID-CCD12>3.0.CO;2-4
- Wakeyama T, Ogawa H, Iida H, et al. Intima-media thickening of the radial artery after transradial intervention. An intravascular ultrasound study. J Am Coll Cardiol. 2003;41(7):1109–1114. doi: 10.1016/s0735-1097(03)00089-5
- Yonetsu T, Kakuta T, Lee T, et al. Assessment of acute injuries and chronic intimal thickening of the radial artery after transradial coronary intervention by optical coherence tomography. Eur Heart J. 2010;31(13):1608–1615. doi: 10.1093/eurheartj/ehq102
- Kamiya H, Ushijima T, Kanamori T, et al. Use of the radial artery graft after transradial catheterization: is it suitable as a bypass conduit? Ann Thorac Surg. 2003;76(5):1505–1509. doi: 10.1016/s0003-4975(03)01018-x
- Kiemeneij F. Prevention and management of radial artery spasm. J Invasive Cardiol. 2006;18(4):159–160.
- Coppola J, Patel T, Kwan T, et al. Nitroglycerin, nitroprusside, or both, in preventing radial artery spasm during transradial artery catheterization. J Invasive Cardiol. 2006;18(4):155–158.
- Ouadhour A, Sideris G, Smida W, et al. Usefulness of subcutaneous nitrate for radial access. Catheter Cardiovasc Interv. 2008;72(3):343. doi: 10.1002/ccd.21645
- Sanmartin M, Cuevas D, Goicolea J, et al. Vascular complications associated with radial artery access for cardiac catheterization. Rev Esp Cardiol. 2004;57(6):581–584. (In Span.) doi: 10.1016/S0300-8932(04)77150-X
- Calvino-Santos RA, Vazquez-Rodriguez JM, Salgado-Fernandez J, et al. Management of iatrogenic radial artery perforation. Catheter Cardiovasc Interv. 2004;61(1):74–78. doi: 10.1002/ccd.10698
- Gunasekaran S, Cherukupalli R. Radial artery perforation and its management during PCI. J Invasive Cardiol. 2009;21(2):E24–26.
- Rigatelli G, Dell'Avvocata F, Ronco F, Doganov A. Successful coronary angioplasty via the radial approach after sealing a radial perforation. JACC Cardiovasc Interv. 2009;2(11):1158–1159. doi: 10.1016/j.jcin.2009.05.026
- Tizon-Marcos H, Barbeau GR. Incidence of compartment syndrome of the arm in a large series of transradial approach for coronary procedures. J Interv Cardiol. 2008;21(5):380–384. doi: 10.1111/j.1540-8183.2008.00361.x
- Kang SS, Labropoulos N, Mansour MA, et al. Expanded indications for ultrasound-guided thrombin injection of pseudoaneurysms. J Vasc Surg. 2000;31(2):289–298. doi: 10.1016/s0741-5214(00)90160-5
- Liou M, Tung F, Kanei Y, Kwan T. Treatment of radial artery pseudoaneurysm using a novel compression device. J Invasive Cardiol. 2010;22:293–295.
- Kanei Y, Kwan T, Nakra NC, et al. Transradial cardiac catheterization: A Review of Access Site Complications. Catheter Cardiovasc Interv. 2011;78(6):840–846. doi: 10.1002/ccd.22978
- Levine GN, Bates ER, Blankenship JC, et al. 2011 ACCF/AHA/SCAI Guideline for Percutaneous Coronary Intervention. A report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines and the Society for Cardiovascular Angiography and Interventions. J Am Coll Cardiol. 2011;58(24):e44–122. doi: 10.1016/j.jacc.2011.08.007
- Sherev DA, Shaw RE, Brent BN. Angiographic predictors of femoral access site complications: implication for planned percutaneous coronary intervention. Catheter Cardiovasc Interv. 2005;65(2):196–202. doi: 10.1002/ccd.20354
- Ben-Dor I, Sharma A, Rogers T, et al. Micropuncture technique for femoral access is associated with lower vascular complications compared to standard needle. Catheter Cardiovasc Interv. 2021;97(70):1379–1385. doi: 10.1002/ccd.29330
- Larena-Avellaneda A, Kölbel T, Carpenter SW, et al. Iatrogene Verletzungen an den Zugangsgefäßen für intravaskuläre Prozeduren. Notfall + Rettungsmedizin. 2017;20:305–314. doi: 10.1007/s10049-017-0287-5
Supplementary files