Effectiveness of intraoperative neurophysiological monitoring during spinal surgery
- Authors: Kuzmina V.А.1, Suynduykov A.R.1, Nikolaev N.S.2,1, Mikhailova I.V.2, Nikolaeva A.V.1
-
Affiliations:
- Federal Center of Traumatology, Orthopedics and Endoprothesis, Cheboksary
- Chuvash State University n.a. I.N. Ulyanov
- Issue: Vol 4, No 4 (2016)
- Pages: 33-40
- Section: Articles
- Submitted: 09.01.2017
- Accepted: 09.01.2017
- Published: 14.12.2016
- URL: https://journals.eco-vector.com/turner/article/view/5893
- DOI: https://doi.org/10.17816/PTORS4433-40
- ID: 5893
Cite item
Abstract
Aim. To assess the application of intraoperative neurophysiological monitoring (IONM) for spinal surgeries in accordance with the Federal Center of Traumatology, Orthopedics and Endoprosthesis (Cheboksary, Russian Federation)
Material and methods. A total of 366 spinal surgeries, Federal Center of Traumatology, Orthopedics and Endoprosthesis from 2009 to 2015. From 2009 to 2013, the wake-up test was used as a control method in 116 (65.9%) cases.
Results. Application of IONM revealed time-dependent risks and facilitated a reduction in the incidence of postoperative neurologic complications by 3-fold (from 2.6% to 0.8%). In the second half of 2013, IONM was introduced for use in clinical practice. Since then, 250 surgeries were performed with IONM. Of these, the wake-up test was required in 9 (3.6%) patients. Clinical implementation of IONM extended the benefits of surgery to patients with severe pathologies. The number of surgeries for congenital pathologies increased by 10-fold (from 1% to 10%) and by 2.6-fold for degenerative diseases. IONM possibility allows control of intraoperative neurologic complications among patients with spinal injuries (5%) and neuro and muscular scoliosis.
Conclusions. The application of IONM minimized the need for the wake-up test and significantly decreased the incidence of neurological complications caused by injury to the spinal cord and spinal roots during execution of spinal manipulations.Full Text
Introduction
Surgery is widely used to treat spinal pathologies, and is the gold standard for correcting spinal deformities [1], but despite improvements in surgical techniques, the risk of neurological complications remains [2]. The introduction of osteotomy procedures, procedures with high correction potential that enable radical elimination of all components of a deformity, minimally invasive surgical techniques, and manipulation of the dural sac and roots to surgical practice does not reduce, and often increases, the risk of iatrogenic neurological complications [3, 4]. The frequency of neurological complications is estimated to range from 0.37% to 10% depending on the method of surgical treatment [5]. The complications can be caused not only by direct mechanical damage, strain or compression of neural structures, but also by disorders of the vascular supply of the spinal cord [6, 7]. Intraoperative neurophysiological monitoring (IONM) helps to prevent such complications [8], and the development low-trauma surgical methods is increasingly relevant, particularly direct lateral interbody fusion (DLIF, XLIF), which is prerequisite for the implementation of IONM [9, 10].
Prior to the implementation of IONM the primary monitoring of neurological complications was the awakening test, also known as the Stagnara wake-up-test, which was first described in 1973. During the wake-up-test, the anesthesiologist interrupts the administration of anesthetics and muscle relaxants, partially wakes the patient to a state in which he/she can execute commands and asks the patient to move the feet. If motor function is not impaired, then anesthesia and surgery continue. Otherwise, an immediate change of surgical approach should be considered. As the test takes 15–20 min and requires repeated administration of anesthetics, application more than once during the intervention is problematic. Consequently, the test is performed after completing the basic corrective manipulations, which significantly reduces its effectiveness because evaluation of the functional integrity of individual muscle groups and nerve trunks is not possible. The use of the wake-up-test does not enable full assessment of neurological status [5]. Intraoperative recovery of consciousness can also lead to complications such as accidental extubation, and air embolism. Pediatric patients are not able to execute the anesthesiologist’s commands during the wake-up-test because of difficulties in verbal communication and their mental status [11]. IONM is becoming the world standard for control of neurological complications during surgical interventions that may affect neural structures [12].
Intravenous anesthesia with propofol as a hypnotic and phentanyl as an analgesic is the most compatible with IONM if combined with ventilation using an oxygen-air inhaled anesthetic mixture, the dose of which is controlled by the maximum alveolar concentration (MAC). If muscle relaxants are necessary, drugs with a medium-duration of action are used. To ensure reliable data and to avoid false-negative results during IONM-guided surgery, the depressor effect of anesthetics on induced activity should be considered. The effects vary from reduction of cortical excitability and delay of axonal transmission in corticospinal tracts to reduction of synaptic transmission at the level of spinal alpha-motoneurons [7, 13, 14].
The effect of general anesthesia and brain sedation were assessed using the bispectral index (BiS) which is calculated from data provided by continuous electroencephalography (EEG). This index enables consistent maintenance of sedation depth, reduces the risk of premature recovery from anesthesia, and reduces the consumption of anesthetics.
In assessing IONM results obtained in pediatric patients, the maturation of the corticospinal tracts should be considered. The functional characteristics of the motor pathways only start to mature at 12–14 years of age. Myelination of the pathways to the muscles of the lower extremities is completed earlier, at 11–12 years of age, and myelination of the pathways to the muscles of the upper extremities is completed by 12–17 years of age. The final maturation of the central motor pathway is completed only in the second decade of life [15, 16]. The small size of muscles in children also requires care when installing the needle electrodes.
Aim. The study aim was to evaluate the use of IONM for spinal surgery at the Federal State Budgetary Institution the Federal Center of Traumatology, Orthopedics and Endoprosthesis (Cheboksary), the Ministry of Health of the Russian Federation.
Materials and Methods
Patient population
The Federal State Budgetary Institution the Federal Center of Traumatology, Orthopedics and Endoprosthesis of the Ministry of Health of the Russian Federation, Cheboksary, Russia (the Center) performed 176 spinal surgeries between 2009 and 2013. Of these, 116 (65.9%) were at risk of neurological complications, and the wake-up-test was used for intraoperative monitoring of the nervous system. The underlying diseases were scoliotic spinal deformity of the third (51%) and fourth (32%) degree, degenerative spinal diseases (10%), kyphotic deformity (6%), and congenital malformations (1%). IONM was introduced in the second half of 2013 and was used in 250 procedures, including 70 men and 180 women from 2 to 71 years of age. Seventeen (6.8%) were pediatric patients younger than seven years of age.
The underlying diseases included congenital spinal malformations in 10% of patients, degenerative diseases 26%, cerebrospinal injuries in 5%, third and fourth degree scoliotic spinal deformity in 22% and 33% of patients, respectively, and kyphosis in 3%. All patients enrolled in the study gave voluntary informed consent for the medical procedures, examinations, surgical interventions, and participation in scientific research before hospitalization (Order No. 125 of 12/30/2013 “On the introduction of forms of documents for legal coverage of the diagnostic and treatment process in the Federal State Budgetary Institution the Federal Center of Traumatology, Orthopedics and Endoprosthesis”).
Intraoperative monitoring
The IONM was performed using the NIM-Eclipse System (Medtronic, Dublin, Ireland) and included six tests.
- A test for muscle relaxants using train-of-four (TOF) monitoring was used to measure the degree of the neuromuscular blockade (NMB). This test enabled avoiding false negative results resulting from paralytic effects of the agents. NMB monitoring was performed by stimulation of a nerve and recording the motor evoked potentials in the innervated muscle.
- Motor evoked potentials (MEP) were recorded in the corresponding muscles during cranial electrotherapy stimulation of the motor cortex. To record MEP, electrodes were placed under the scalp along lines spaced a finger width apart and drawn forward (i.e., towards the nose) from points C3 and C4 and the corresponding projections of the motor cortex.
- Control of proximity to the nerve (N. Proxy) allowed monitoring the correct channel of the pedicle screw. A loop was attached to the surgeon’s instrument, and a 1–12 mA electrical stimulus was applied. When the instrument passed over the vertebral arch, an isoline was recorded on the device monitor. With proximity of the conductor to the neural structures, an EMG response appeared and the amplitude increased with the strength of the stimulus. The surgeon then changed the direction of instrument movement until the EMG response disappeared.
- The pedicle screw integrity test verified the accuracy of pedicle screw installation during spinal fixation procedures. An electrical stimulus was applied using a flexible bougie, with the instrument in the Screw Integrity mode. The absence of an EMG response or its appearance following a strong stimulus indicated the correct mounting of the screw in the pedicle and the absence of defects of the pedicle canal walls. The presence of the EMG response was indicated proximity to the neural structures, because of lack of bone tissue in the pedicle. This was often seen at the deformity apex on the concave side and occurred because of the anatomical and morphological features of the vertebrae in this region. On registration of a high-amplitude EMG response, control of the screw position was performed using an electro-optical converter (EOC). If the screw was not in the correct position, it was removed if it did not carry a strategic load or the trajectory was changed, in some cases the pedicle screw was replaced by another fixing element such as a hook, or tape.
- The N. Root test of the spinal roots was done using automatic electrical stimulation. Intact nerve roots that innervated the relevant muscles produced an EMG response after receiving a stimulus of < 2 mA.
- For EMG recording, needle electrodes were mounted on the muscles corresponding to the myotomes innervated by the spinal cord segments at the level of the surgical intervention. The electrodes were mounted prior to draping the patient and creating the sterile surgical field.
Surgery
Premedication consisted of atropine, seduxen (relanium) or dormicum, and diphenylhydramine administered intravenously five min prior to general and endotracheal anesthesia. Patients were intubated orotracheally intravenous administration of the muscle relaxant suxamethonium chloride. After intubation, the patients were transferred to mechanical ventilation. Phentanyl, propofol, sevoflurane in a MAC of 0.3% to 0.5% were administered as supportive anesthesia.
The surgical procedures involved access to the dorsal structures of the spine by both open and minimally invasive techniques using specialized retractor systems. The dorsal bony structures of the spine were skeletonized, and the hardware support elements (pedicle screws) were installed. Depending on the procedure, the next stage was decompression, osteotomy, or corrective procedures. The achieved result was stabilized by the installation of a spinal system. The dorsal phase of the surgical intervention was completed by the formation of spondylodesis along the hardware, the wound layers were tightly closed. Depending on the stage of the surgery, the muscle relaxant (TOF) test, test of nerve proximity, the proximity screw test, and EMG were performed. The N. Root test was performed for the patients with degenerative spine diseases. During surgery, transcranial stimulation of the motor cortex (MEP) was used to monitor the status of the pyramidal tract.
Results and Discussion
Of the 116 patients surgical patients monitored with the wake-up-test, three (2.6%) were not able to move their legs during recovery, which indicated the development of intraoperative neurological complications. Postoperative therapy resulted in an increase in strength and sensitivity in the legs, and complete recovery was achieved by all three patients.
With implementation of IONM, the patient population changed because the underlying diseases that were treated included increasingly complex (Fig. 1).
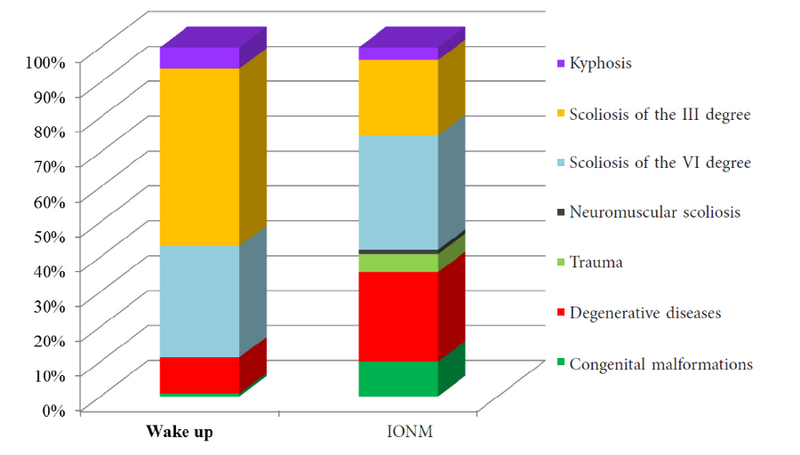
Fig. 1. The nature of the underlying disease of the patients with surgical treatment with intraoperative neuromonitoring and the wake-up-test on the nosology (%)
Since the second half of 2013, the number of surgeries for congenital pathologies increased by 10 times, or from 1% to 10% of cases, and increase by 2.6 times the degenerative diseases increased by 2.6 times. Control of intraoperative neurological complications became possible in patients with spinal traumas (5%) and neuromuscular scoliosis. The number of patients with surgical treatment of the spinal kyphosis decreased from 6% to 3%, and the wake-up-test was performed only in patients with Scheuermann’s disease. With the availability of IONM, more complex kyphoses were treated, including osteodysplasia and Marie–Strumpell disease, and patients with Schwab group 3 or 4 deformities requiring osteotomies, all of which are accompanied by a high risk of neurological complications. In all 250 patients, IONM began with the MEP test, and the dynamic evaluations were performed throughout surgery (Table 1).
Table 1. Results of the test of the MEP test
Motor evoked potentials | Number of patients (n) | Proportion, % |
Norm | 204 | 81.6 |
Absence | 15 | 6 |
Initially normal value of the motor evoked potentials with the decrease in angle by more than 70% during the surgery | 14 | 5.6 |
Motor evoked potentials in pediatric patients under seven years of age | 17 | 6.8 |
Total | 250 | 100 |
In most patients, normal MEP values were recorded initially and during the surgery, which indicated the integrity of motor pathways, and did not require changes in the surgical procedure. The absence of the MEP occurred in 15 patients with the failed back surgery, and the consequences of cerebrospinal injury, fourth degree kyphoscoliotic deformity of the thoracic spine, congenital malformation of the spine with neurological disorders (paraparesis and plegia). In the postoperative period, the neurological symptoms persisted, which indicated deep motor pathway lesions. Worsening of neurological symptoms occurred in two (0.8%) patients, and was considered as a postoperative complication. During the follow-up treatment improvement was seen (Table 2).
Table 2. Comparative characteristics of neurological complications of patients with the wake-up-test and with intraoperative neuromonitoring
№ | Gen-der | Age | Underlying disease | Amount of deformity, Cobb angle, ° | Neurological complication | Control test |
1 | m | 16 | Juvenile idiopathic scoliosis of the 4 degree, Lenke type 3a | 160 | Vascular myelopathy with flail legs, the pain sensitivity disorder on the conduction type from the level of D5 | Wake-up-test |
2 | m | 18 | Juvenile idiopathic scoliosis of the 4 degree, Lenke type 3b | 130 | Flail legs and pain sensitivity disorder on the conduction type from the level of D5 | Wake-up-test |
3 | m | 15 | Left-sided lower thoracic scoliosis of the 4 degree due to osteochondrodystrophy with the upper thoracic compensatory curve | 96 | Moderate right-sided flaccid monoparesis | Wake-up-test |
4 | m | 19 | Idiopathic infantile right-sided thoracic kyphoscoliosis of the 4 degree, complicated by lower spastic paraparesis 2–3 degree, with the segmental sensory disorders due to the spinal deformity, myelopathy | 136 | Lower spastic paraplegia with total disorder of the superficial and deep sensitivity from the level TH9–TH10 and the pelvic disorders on the type of retention of urine and stool | IONM |
5 | m | 3 | Congenital malformation of the spine due to the damage of the formation, fusion of the vertebrae Th12. Kyphosis at the thoracolumbar transition level with the dislocation L1, the spinal stenosis. The neutral malformation of the cervical spine. The lower spastic paraparesis of the moderate severity with the reference on the right. Residual encephalopathy, arrested psychoverbal development | 69 | Lower flaccid paraplegia | IONM |
Decreases of up to 70% in motor response were seen in 14 patients with initially normal MEP values, and as the cause in each case was transient disturbance of motor nerve conduction, the initial operative plan was changed. Prompt administration of glucocorticoids, diuretics, and a decrease in corrective actions in five patients, resulted in normalization of MEP and allowed continuing the surgery. The wake-up-test was required in nine (3.6%) patients and was positive in all. All nine patients in that group had a severe deformity of the spine that required complex surgical correction. In the postoperative period, neurologic complications were not observed. It should be noted that prior to the introduction of IONM the wake-up-test was performed in 65.9% patients.
Pediatric patients under seven years of age with surgical treatment of the spine and spinal cord malformations were evaluated separately. Initially, normal MEP amplitudes were recorded in the abdominal rectus and oblique abdominal muscles. Reduced amplitudes, in some cases to the isoline, were recorded in the muscles of femur and tibia. Reduction of MEP amplitude was associated with the degree of maturation of the corticospinal tract and the depressor effect on the motor cortex of the Sevoran inhalation anesthetic. Withdrawal of Sevoran resulted in the restoration of normal MEP. The BiS optimal index for correct estimation of the IONM parameters was 33–50.
Given the hands-free method used by surgeons to position transpedicular support elements on anatomical landmarks, N. Proxy test of proximity to neural structures was an important element. The test results indicated that the surgeon’s positioning needed correcting. In addition to the MEP test, The N. Root test was used for the 34 patients with degenerative diseases of the spine. At the baseline and at completion of surgery, normal MEP levels in these patients confirmed the integrity of the corticospinal tract. The N. Root test allowed determining the distance of the lesion from the spinal roots. The test results were correlated with the clinical picture. The intact roots responded to stimuli of up to 2 mA in 22 (64%) patients, in eight patients (23.5%) with paresis the required stimulus strength increased to 6–10 mA. In the four patients (12.5%) with plegia no response was observed even with a maximum stimulus strength of 15 mA. The screw integrity test was performed after installing the pedicle screws. In our practice, an EMG response was obtained in three patients (1.2%) following a stimulus of 6 mA, which indicated the dangerous proximity of the screw to the neural structures and required use of the EOC to correct. A similar situation was observed at the deformity apex on the concave side and was the result of the anatomical and morphological features of the vertebrae in that region. In one patient, an incorrectly positioned screw was removed as it did not carry a strategic load. In the second patient, the conduction trajectory was changed, and in the third patient the pedicle screw was replaced by another locking element (tape).
Summary and Conclusions
The success of surgical intervention depends on the coordinated work of all members of the surgical team, the surgeon, the anesthesiologist, the neurophysiologist. The introduction of IONM enabled to significant expansion of the patient population because of the ability to treat more complex pathologies. Thus, 2.6 times increase in the use of IONM in the patients with degenerative spine diseases was associated with the ability to control the extent of lesions of the nerve roots. IONM, with the use of the NIM-EclipseSystem, allows multiple intraoperative assessments of the functional integrity of motor pathways and spinal roots from the time of the establishing the channel for the pedicle screw until the completion of surgery. The use of wake-up-tests is minimized, which is significant because performing them than once during the surgery is impossible, or dangerous for the patient. Many complex surgical procedures for congenital malformations, severe spine deformities, osteotomy of type 3 and 4 deformities is not possible without IONM. It should be noted that the IONM in pediatric practice requires an individual approach. The small size of the muscles and the depressor effect of inhalation anesthetics should be considered. Application of IONM enabled prompt identification of risks and reduced postoperative neurological complications by three times, from 2.6% to 0.8%). It allowed expanding the range of surgical procedures performed improving the quality of treatment of patients with spinal disorders.
Author Contributions
Kuzmina V.A. was responsible for the collection and processing of the materials, data analysis, and writing the manuscript. Syundyukov A.R. was responsible for the collection and processing of the materials, data analysis, writing the manuscript, and the concept and design of the study. Mikhailova I.V. was responsible for advising on and writing the manuscript. Nikolaeva A.V. was responsible for obtaining and analyzing data, writing the manuscript, and the concept and design of the study.
Funding and Conflict of Interest
The work was performed with the support of the I.N. Ulyanov Chuvash State University. The authors declare the absence of apparent or potential conflicts of interest associated with the publication of this article.
About the authors
Valentina А. Kuzmina
Federal Center of Traumatology, Orthopedics and Endoprothesis, Cheboksary
Author for correspondence.
Email: doctorkuzmina@rambler.ru
MD, doctor of functional diagnostics Russian Federation
Airat R. Suynduykov
Federal Center of Traumatology, Orthopedics and Endoprothesis, Cheboksary
Email: sndk-ar@yandex.ru
MD, orthopedic and trauma surgeon children’s trauma and orthopedic department Russian Federation
Nikolay S. Nikolaev
Chuvash State University n.a. I.N. Ulyanov; Federal Center of Traumatology, Orthopedics and Endoprothesis, Cheboksary
Email: fc1@orthoscheb.com
MD, PhD, professor, Head of the chair of traumatology, orthopedics and emergency medicine of the Chuvash State University n.a. I.N. Ulyanov. Head doctor of the Federal Center of Traumatology, Orthopedics and Endoprothesis, Cheboksary Russian Federation
Irina V. Mikhailova
Chuvash State University n.a. I.N. Ulyanov
Email: fake@eco-vector.ru
MD, PhD, assistant professor of the chair of functional and laboratory diagnostics Russian Federation
Alena V. Nikolaeva
Federal Center of Traumatology, Orthopedics and Endoprothesis, Cheboksary
Email: orlova.alena@mail.ru
MD, PhD, doctor of metodical department Russian Federation
References
- Михайловский М.В., Фомичев Н.Г. Хирургия деформаций позвоночника. — Новосибирск, 2002. — 428 с. [Mikhailovskiy MV, Fomichev NG. Spinal Deformities Surgery. Novosibirsk; 2002. 428 p. (In Russ.)].
- Удалова И.Г., Михайловский М.В. Неврологические осложнения в хирургии сколиоза // Хирургия позвоночника. — 2013. — № 3. — С. 38–41. [Udalova IG, Mikhailovskiy MV. Neurological Complications in Scoliosis Surgery. Spine surgery. 2013;(3):38-41. (In Russ.)].
- Wright N. XLIF - the United States Experience 2003-4. International Meeting on Advanced Spinal Techniques Banff Alberta. Canada; 2005.
- Anenbach JD, Lenke LG, Bridwell KH, et al. Major complications & companison between 3-column osteotomy techniquses in 105 consecutive spinal deformity procedures. Spine. 2015;14:198-210.
- Новиков В.В., Новикова М.В., Цветовский С.Б., и др. Профилактика неврологических осложнений при хирургической коррекции грубых деформаций позвоночника // Хирургия позвоночника. — 2011. — № 3. — С. 66–74. [Novikov VV, Novikova MV, Tsvetkovskiy SB, et al. Prevention of Neurological Complications in Severe Spinal Deformities Surgery. Spine surgery. 2011;(3):66-74. (In Russ.)]
- Фоменко М.В., Голубев Г.Ш., Лака А.А., Купленский А.Э. Результаты хирургического лечения детей с прогрессирующим идиопатическим сколиозом // Хирургия позвоночника. — 2010. — № 2. — С. 35–40. [Fomenko MV, Golubev GSh, Laka AA, Kuplenskiy AE. Results of Surgical Treatment of Progressive Idiopathic Scoliosis in Children. Spine Surgery. 2010;(2):35-40. (In Russ.)].
- Хабиров Ф.А. Руководство по клинической неврологии позвоночника. — Казань: Медицина, 2006. — 518 с. [Khabirov FA. Guidelines for Clinical Neurology of Spine. Kazan: Medicine; 2006. 518 p. (In Russ.)]
- Щекутьев Г.А. Нейрофизиологические исследования в клинике НИИ нейрохирургии им. Н.Н. Бурденко. — М.: Антидор, 2001. — 233 с. [Schekutev GA. Neurophysiological Researches in Clinic Research Institute of Neurosurgery named after N.N. Burdenko. Moscow: Antidor; 2001. 233 p. (In Russ.)]
- Tohmeh AG, Rodgers WB, Peterson MD. Dynamically evoked, discrete-threshold electromyography in the extreme lateral interbody fusion approach. J Neurosurg Spine. 2011;14(1):31-37. doi: 10.3171/2010.9.spine09871.
- Wright N. XLIF-the United States Experience 2003-4. International Meeting on Advanced Spinal Techniques Banff Alberta. Canada, 2005.
- Хить М.А., Колесов С.В., Колбовский Д.А., Морозова Н.С. Роль интраоперационного нейрофизиологического мониторинга в предотвращении развития послеоперационных неврологических осложнений в хирургии сколиотической деформации позвоночника // Нервно-мышечные болезни. — 2014. — № 2. — С. 36–41. [Hit MA, Kolesov SV, Kolbovky DA, Morozova NS. The role of intraoperativeneurophysiological monitoring in prevention of postoperative neurological complications in scoliotic spinal deformation surgery. Neuromucular diseases. 2014;(2):36-41. (In Russ.)]
- Огурцова А.А. Интраоперационный нейромониторинг в нейрохирургии: сборник трудов. — М., 2013. — 74 с. [Ogurtsova AA. Intraoperative neuromonitoring in Nneurosurgery: Collection of Studies. Moscow; 2013. 74 p. (In Russ.)]
- Aatif M, Husain A. Practical Approach to Neurophysiologic Intraoperative Monitoring. New York; 2008. P. 882-898.
- The LAFS (Los Angeles Anesthesia Friendliness Scale). 2001.
- Никитин С.С., Куренков А.Л. Магнитная стимуляция в диагностике и лечении болезней нервной системы. Руководство для врачей. — М.: САШКО, 2003. — 378 с. [Nikitin SS, Kurenkov AL. Magnetic stimulation in the diagnosis and treatment of diseases of the nervous system. Guidelines for doctors. Moscow: Sashko; 2003. 378 р. (In Russ.)]
- Muller K, Kass-Iliyya F, Reitz M. Ontogeny of ipsilateral corticospinal projections: a developmental study with transcranial magnetic stimulation. Ann Neurol. 1997;42(5):705-711. doi: 10.1002/ana.410420506.
Supplementary files








